Improving haloalkane dehalogenase thermostability by +20 °C while enhancing activity and expression
Scientists at Cradle optimized a haloalkane dehalogenase for thermostability and expression while preserving activity across two rounds. The best candidate achieved a melting temperature increase of +20.0 °C to 65.1 °C, 2.06× improved expression, and 1.29× enhanced activity.

Daniel

Daniel
Modality | Enzyme (haloalkane dehalogenase, DhaA) |
Target | DhaA (accession Q53042) |
Properties optimized | Thermostability, expression, activity |
Rounds | 2 |
Candidates per round | 96 |
Key result | ΔT_m +20.0 °C (to 65.1 °C), 2.06× expression, 1.29× activity |
Partner | Internal |
Data availability | Detailed in CRADLE-1 whitepaper |
Context
Haloalkane dehalogenases catalyze the cleavage of carbon-halogen bonds and have applications in bioremediation, biosensing, and protein labeling. Industrial and biotechnological applications often require enzyme operation at elevated temperatures or under denaturing conditions, making thermostability a critical property for practical deployment. Simultaneously maintaining or improving catalytic activity and expression during stability engineering represents a common but challenging multi-objective problem in enzyme engineering.
Challenge
The template sequence exhibited a melting temperature of 45.1 °C—substantially below the temperatures encountered in many industrial processes. Previous attempts to engineer thermostable variants often resulted in reduced catalytic activity or expression, reflecting the typical trade-offs in enzyme engineering where stability-enhancing mutations can rigidify the protein and impair function.
The optimization required balancing three properties with potential antagonistic relationships: increasing thermostability (which often reduces flexibility needed for catalysis), maintaining or improving expression (which can be disrupted by destabilizing mutations), and preserving or enhancing activity (which requires maintaining the catalytic mechanism).

Meet the Cradle wet lab
Discover why we, unlike other AI companies, run our own wet lab in Amsterdam to build better models.
Approach
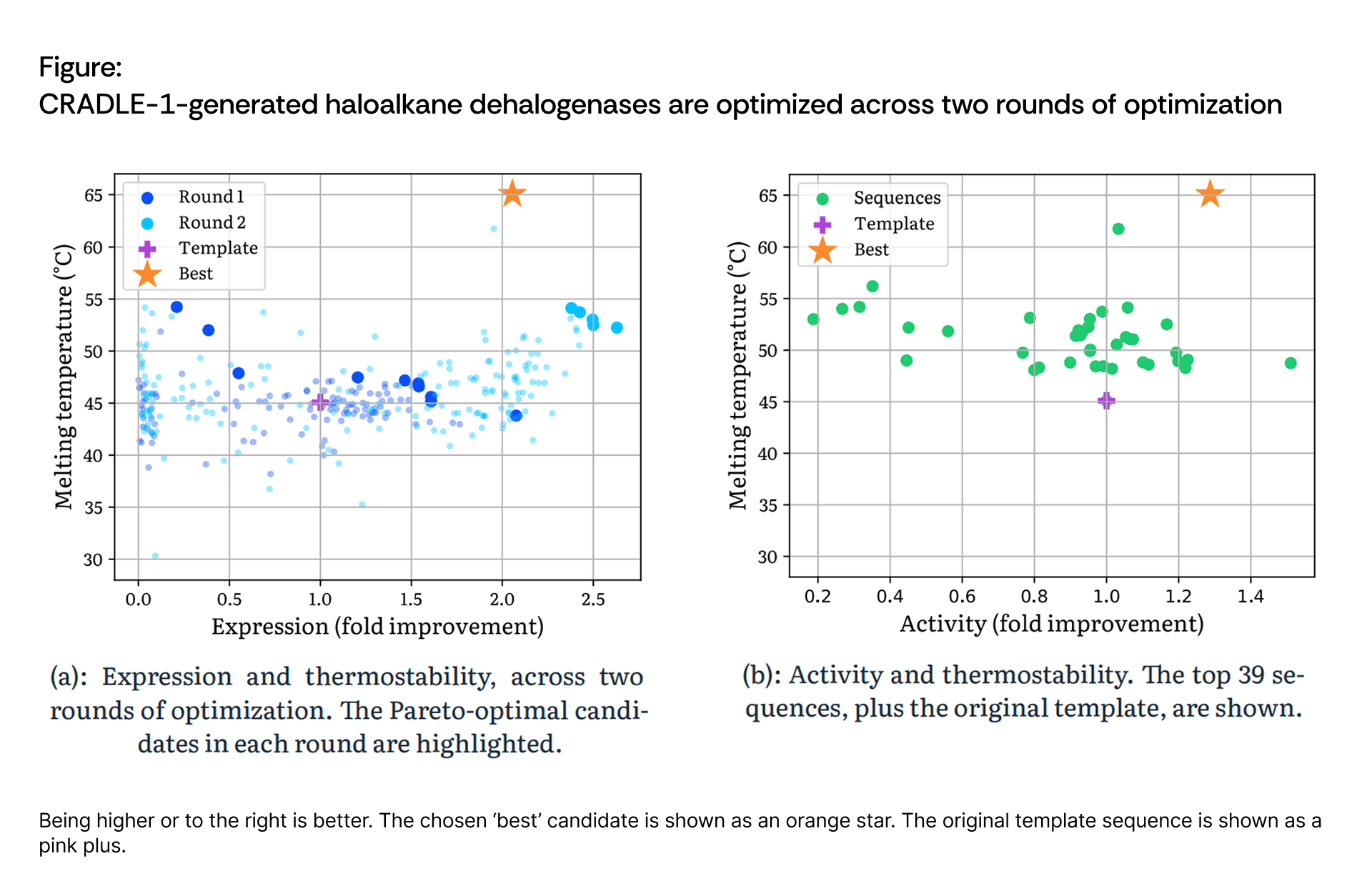
Scientists at Cradle performed two rounds of Cradle optimization with 96 candidates each. Round 1 operated in zero-shot mode for diversification. Round 2 was configured with thermostability as the primary objective while setting expression as a relative constraint to ensure improvement over the template.
After observing that Round 1 generated some poorly expressing variants, the Round 2 strategy explicitly prioritized expression recovery alongside continued thermostability gains. Among the Round 2 candidates with improved expression over the template, the top 39 by thermostability were selected for activity assaying to identify variants that successfully maintained or enhanced catalytic function.
Results
The optimization successfully achieved substantial gains across all three properties simultaneously:
Property | Template | Best CRADLE-1 candidate | Improvement |
T_m | 45.1 °C | 65.1 °C | +20.0 °C |
Expression | 1× | 2.06× | 2.06× |
Activity | 1× | 1.29× | 1.29× |
The +20 °C increase in melting temperature represents a 44% improvement in thermal stability, substantially expanding the operational temperature range for this enzyme. Critically, this improvement came alongside enhanced rather than diminished activity—the best candidate exhibited 1.29× higher catalytic turnover than the template.
The 2.06-fold expression improvement addresses a common manufacturability bottleneck. Higher-expressing variants reduce production costs and enable applications requiring large quantities of enzyme.
The Pareto frontier plots across Rounds 1 and 2 demonstrate progressive optimization of the thermostability-expression trade-off, with Round 2 successfully recovering expression while maintaining the thermostability gains achieved in Round 1. Among the 39 high-thermostability candidates selected for activity testing, multiple variants exhibited both improved expression and enhanced activity, demonstrating that CRADLE-1 identified regions of sequence space where all three properties could be simultaneously improved.
The final best sequence differs from the template by only 6 mutations, demonstrating that Cradle can achieve significant property gains with minimal structural perturbation.
What this means
This result demonstrates that Cradle can navigate complex multi-property enzyme optimization problems where conventional wisdom suggests inherent trade-offs between stability and function. Thermostability, activity, and expression are typically decorrelated, meaning improvements in one often come at the cost of another. Cradle was able to walk this tightrope, identifying sequences that improved all three properties by navigating the complex trade-off landscape.
For industrial enzyme applications, the ability to achieve these improvements in just two rounds (compared to typical directed evolution campaigns requiring many more rounds) substantially accelerates development timelines for biocatalysts operating under harsh conditions.
Methods note
Cradle employed evotuning on the DhaA evolutionary alignment followed by supervised fine-tuning on Round 1 wet lab data for Round 2 generation. Thermostability was measured via differential scanning fluorimetry, expression via LabChip quantification of E. coli lysates, and activity via a fluorescence-based spectrophotometric assay monitoring cleavage of 4-bromomethyl-6,7-dimethoxycoumarin. Full protocols are detailed in the Cradle whitepaper.
Modality | Enzyme (haloalkane dehalogenase, DhaA) |
Target | DhaA (accession Q53042) |
Properties optimized | Thermostability, expression, activity |
Rounds | 2 |
Candidates per round | 96 |
Key result | ΔT_m +20.0 °C (to 65.1 °C), 2.06× expression, 1.29× activity |
Partner | Internal |
Data availability | Detailed in CRADLE-1 whitepaper |
Context
Haloalkane dehalogenases catalyze the cleavage of carbon-halogen bonds and have applications in bioremediation, biosensing, and protein labeling. Industrial and biotechnological applications often require enzyme operation at elevated temperatures or under denaturing conditions, making thermostability a critical property for practical deployment. Simultaneously maintaining or improving catalytic activity and expression during stability engineering represents a common but challenging multi-objective problem in enzyme engineering.
Challenge
The template sequence exhibited a melting temperature of 45.1 °C—substantially below the temperatures encountered in many industrial processes. Previous attempts to engineer thermostable variants often resulted in reduced catalytic activity or expression, reflecting the typical trade-offs in enzyme engineering where stability-enhancing mutations can rigidify the protein and impair function.
The optimization required balancing three properties with potential antagonistic relationships: increasing thermostability (which often reduces flexibility needed for catalysis), maintaining or improving expression (which can be disrupted by destabilizing mutations), and preserving or enhancing activity (which requires maintaining the catalytic mechanism).

Meet the Cradle wet lab
Discover why we, unlike other AI companies, run our own wet lab in Amsterdam to build better models.
Approach
Scientists at Cradle performed two rounds of Cradle optimization with 96 candidates each. Round 1 operated in zero-shot mode for diversification. Round 2 was configured with thermostability as the primary objective while setting expression as a relative constraint to ensure improvement over the template.
After observing that Round 1 generated some poorly expressing variants, the Round 2 strategy explicitly prioritized expression recovery alongside continued thermostability gains. Among the Round 2 candidates with improved expression over the template, the top 39 by thermostability were selected for activity assaying to identify variants that successfully maintained or enhanced catalytic function.
Results
The optimization successfully achieved substantial gains across all three properties simultaneously:
Property | Template | Best CRADLE-1 candidate | Improvement |
T_m | 45.1 °C | 65.1 °C | +20.0 °C |
Expression | 1× | 2.06× | 2.06× |
Activity | 1× | 1.29× | 1.29× |
The +20 °C increase in melting temperature represents a 44% improvement in thermal stability, substantially expanding the operational temperature range for this enzyme. Critically, this improvement came alongside enhanced rather than diminished activity—the best candidate exhibited 1.29× higher catalytic turnover than the template.
The 2.06-fold expression improvement addresses a common manufacturability bottleneck. Higher-expressing variants reduce production costs and enable applications requiring large quantities of enzyme.
The Pareto frontier plots across Rounds 1 and 2 demonstrate progressive optimization of the thermostability-expression trade-off, with Round 2 successfully recovering expression while maintaining the thermostability gains achieved in Round 1. Among the 39 high-thermostability candidates selected for activity testing, multiple variants exhibited both improved expression and enhanced activity, demonstrating that CRADLE-1 identified regions of sequence space where all three properties could be simultaneously improved.
The final best sequence differs from the template by only 6 mutations, demonstrating that Cradle can achieve significant property gains with minimal structural perturbation.

What this means
This result demonstrates that Cradle can navigate complex multi-property enzyme optimization problems where conventional wisdom suggests inherent trade-offs between stability and function. Thermostability, activity, and expression are typically decorrelated, meaning improvements in one often come at the cost of another. Cradle was able to walk this tightrope, identifying sequences that improved all three properties by navigating the complex trade-off landscape.
For industrial enzyme applications, the ability to achieve these improvements in just two rounds (compared to typical directed evolution campaigns requiring many more rounds) substantially accelerates development timelines for biocatalysts operating under harsh conditions.
Methods note
Cradle employed evotuning on the DhaA evolutionary alignment followed by supervised fine-tuning on Round 1 wet lab data for Round 2 generation. Thermostability was measured via differential scanning fluorimetry, expression via LabChip quantification of E. coli lysates, and activity via a fluorescence-based spectrophotometric assay monitoring cleavage of 4-bromomethyl-6,7-dimethoxycoumarin. Full protocols are detailed in the Cradle whitepaper.
Recent posts
Subscribe and get new posts and updates from Cradle straight to your inbox.
Follow Cradle
Built with ❤️ in Amsterdam & Zurich
Follow Cradle
Built with ❤️ in Amsterdam & Zurich
Follow Cradle